How To Determine Limiting Reactant With Moles
The limiting reagent depends on the mole ratio not on the masses of the reactants present. Determine which reactant is limiting by dividing the number of moles of each reactant by its stoichiometric coefficient.

Limiting Reactant Practice Problem Science Sciencewithtylerdewitt Tylerdewitt Tutor Sciencehelp School Help Online School Chemistry
Calculate the number of moles of each reactant present.

How to determine limiting reactant with moles. This reactant generally determines when the reaction will stop. Once you find the moles only convert one of them to the moles of the other reactant. Consider for example burning propane in a grill.
What is a Limiting Reactant. Remember this is determined based on the mole ratio of H2 and H2O which is 22 the coefficients in front of each molecule. In our case the limiting reactant is oxygen and the amount of product NO produced from it is 25 moles.
Thus the theoretical yield for the reaction is 25 moles. Calculate the number of moles of each reactant by multiplying the volume of each solution by its molarity. This chemistry video tutorial provides a basic introduction of limiting reactants.
To find out the limiting reagent you need to find the amount of product that can be made with respect to each reactant involved. Divide the actual number of moles of each reactant by its stoichiometric coefficient in the balanced. Divide the actual number of moles of each reactant by its stoichiometric coefficient in the balanced chemical equation.
It explains how to identify the limiting reactant given the mass in grams. Remember to use the molar ratio between the limiting reactant and the product. In simpler words it is the amount of product produced from the limiting reactant.
The reason for using a limiting reactant is that the elements and compounds react with each other in a balanced chemical equation according to the mole ratio between them. If you define limiting reagent it is a reactant in a chemical reaction which determines the amount of product which is produced. To identify the limiting reactant calculate the number of moles of each reactant present and compare this ratio to the mole ratio of the reactants in the balanced chemical equation.
5272 mol of TiCl 4 and 823 mol of Mg. The exact amount of reactant which will be needed to react with another element can be calculated from the reaction stoichiometry. To identify the limiting reactant calculate the number of moles of each reactant present and compare this ratio to the mole ratio of the reactants in the balanced chemical equation.
The propane and oxygen in. Use mole ratios to calculate the number of moles. The way I calculate the limiting reactant is by first finding the amount of moles are in the reactants given.
Use the mole ratios from the balanced chemical equation to convert from moles of limiting reactant to moles of product and then use the gram-mole conversion factor from the periodic table to. For example if you had a equation of 2h22o2----2h202 find the moles of h2 through o2 by multiplying the moles of o2 found to the moles of h2 over. This reactant is known as the limiting reactant.
Once the limiting reactant is determined the moles of product can be determined. According to the balanced chemical equation every 2 moles of H2 will yield 2 moles of H2O. A If the calculated MOLES NEEDED is greater than the MOLES HAVE for a given reactant then that reactant is the limiting reagent.
Compare Moles Have with Moles Used to determine limitingexcess reagent. Causey shows you how to find the limiting reactant reagent and the maximum product from a chemical equation using stoichiometry. Suppose you have the following chemical equation and you are asked to find the limiting reactant if the amount of sodium is 25g and that of chlorine is 40g.
In order to determine the limiting reactant we need to determine which of the reactants will give less product. Determine the number of moles of each reactant. Here is a simple and reliable way to identify the limiting reactant in any problem of this sort.
If we only have x moles of a reactant we can only expect y moles of product. The actual yield is the amount of end product obtained upon experimentation. Often it is straightforward to determine which reactant will be the limiting reactant but sometimes it takes a few extra steps.
In a given stoichiometry problem you will use this reactant to determine. The reactant that would produce the smallest amount of product is the limiting reagent. The steps to determine the limiting reagent or the limiting reactant is as follows.

Need To Calculate The Limiting Reactant Of A Chemical Reaction Chemical Reactions Chemical Chemical Equation

How To Calculate An Empirical Formula Chemistry Worksheets Teaching Chemistry Chemistry Help

Stoichiometry Limiting Reagent Stoichiometry Chemistry Chemistry School Help

What Is Stoichiometry And Why Is It Used In Chemistry A Plus Topper Whatisstoichiometry Stoichiometry Chemistry Chemistry Chemistry Worksheets

Amazing Chemical Chemistry Lessons Chemistry Worksheets Teaching Chemistry

Chemistry 12 3 Limiting Reagent And Percent Yield Chemistry Percents Pure Products

Equilibrium 2 Calculating Equilibrium Teaching Chemistry How To Learn Chemistry Chemistry Lessons

How To Find The Limiting Reactant And Percent Yield Chemistry Notes Chemistry Lecture Lectures Notes

Mole Conversions Made Easy How To Convert Between Grams And Moles Youtube Scientific Skills Teaching Chemistry College Words

Limiting Reactant Example Problem 1 Chemistry Problem Fuel Cells
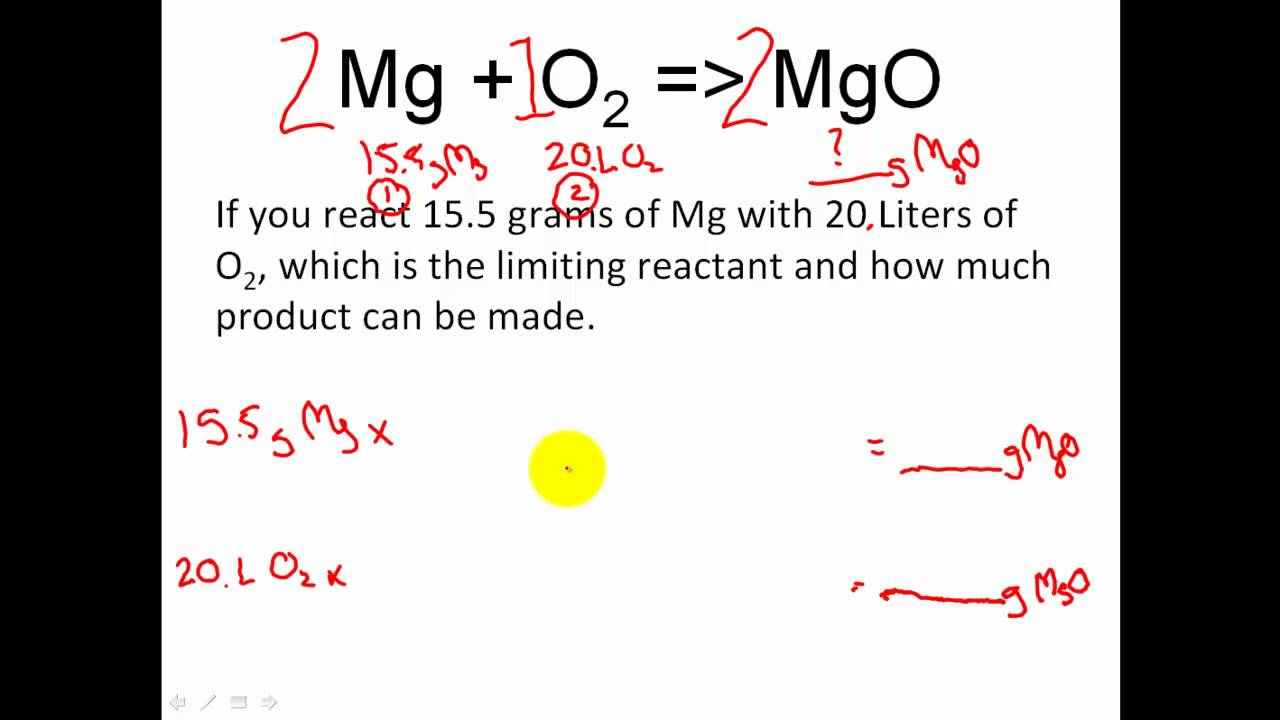
Stoichiometry Limiting Reactant Excess Reactant Stoichiometry Moles Module 6 Learning Psychology Apologia Chemistry School Work

Excess Reactant Unit 1 Stoichiometry Com Imagens Quimica

How To Calculate Percent Yield In Chemistry Teaching Chemistry Chemistry Physical Chemistry

Balance Different Types Of Reactions With A Simple Yet Efficient Method To Help Avoid All The Confusion M Chemistry Worksheets Teaching Chemistry Mcat Study

Limiting Reactants And Percent Yield Apologia Chemistry Chemistry Chemistry Class

Percent Error Worksheet Answer Key Fresh Percent Yield Worksheet Super Teacher Worksheets Chemistry Worksheets Chemistry

An In Depth Introduction To Balancing Redox Equations Complete With A Step By Step Guide To Make Sure Nothing Is Missed In The P Redox Reactions Mcat Chemistry

Limiting Reagent Chemistry Tutorial Youtube Chemistry Tutorial School

Post a Comment for "How To Determine Limiting Reactant With Moles"