How To Determine Limiting Reactant Given Moles
Determine the balanced chemical equation for the chemical reaction. Limiting reactant is important as it determines when the reaction stops and it also controls the amount of product made.

Introduction To Limiting Reactant And Excess Reactant Science Sciencewithtylerdewitt Tylerdewitt Tu Chemistry Help Apologia Chemistry High School Chemistry
Given moles of multiple reactants determine which is limiting and calculate the moles of product formed.

How to determine limiting reactant given moles. Pick a reactant and calculate how much product you can make assuming excess of. Follow Report by Arpatelzoya4449 15042019 Log in to add a. Identify what is given and what is asked for.
Finally determine the moles of product formed from balanced equation and grams of product formed from periodic table gram-mole conversion factor. In order to determine the limiting reactant we need to determine which of the reactants will give less product. Then use the balanced equation to calculate the number of moles of sulfur that would be needed to react with the number of moles of silver present.
Calculate the yield of each reactant. Calculate the theoretical yield of a reaction and calculating the percent yield of a reaction. Determine which reactant is limiting by dividing the number of moles of each reactant by its stoichiometric coefficient in the balanced chemical equation.
10 points How to find the limiting reactant given moles. How to find the limiting reactant given moles. Whichever value is smallest is the limiting reactant.
How To Determine Limiting Reactant With Moles. Calculate the mole ratio from the given. Compare moles have with moles needed Since moles needed of N 2 165 moles exceeds the moles have 0357 moles N 2 is the limiting reagent.
How to Solve Moles-to-Moles Limiting Reactant Problems Step 1. Calculate the limiting reactant for 1 mol n2o4 we need 2 moles n2h4 to produce 3 moles n2 and 4 moles h2o n2o4 is the limiting reactant. To determine which reactant is the limiting reactant first determine how much product would be formed by each reactant if all the reactant was consumed.
Remember this is determined based on the mole ratio of H2 and H2O which is 22 the coefficients in front of each molecule. Then determine the limiting reactant using mole ratios from the balanced equation. According to the balanced chemical equation every 2 moles of H2 will yield 2 moles of H2O.
The reactant that forms the least amount of product will be the limiting reactant. Answer If youre given the moles present of each reactant and asked to find the limiting reactant of a certain reaction then the simplest way to find which is limiting is to divide each value by that substances respective coefficient in the balanced chemical equation. A value less than the ratio means the top reactant is the limiting reactant.
Determine the number of moles of each reactant. First determine the moles of reactants initially present using the molarity conversion factor. Kindle File Format How To Determine Limiting Reactant When Given Moles Limiting Reactant and Percent-Lifeliqe 2019 This lesson plan covers analyzing chemical reactions in order to determine limiting reactants and excess reactants including calculating the amount of excess reactant.
Ask for details. Use mole ratios to calculate the number of moles of product that can be formed from the limiting reactant. Get the answers you need now.
Use the atomic masses of ceAg and ceS to determine the number of moles of each present. Multiply the number of moles of the product by its molar mass to obtain the corresponding mass of product. 279 Since our value is less than the ideal ratio the top reactant is the limiting reactant.
Compare this result to the actual number of moles of sulfur present. Formula to calculate limiting reactant. Convert all given information into moles most likely through the use of molar mass as a conversion factor.
0357 moles N 2 X 2 moles NH 3 1 mole N 2 X 170 g NH 3 1 mole NH 3 121 g NH 3 1 3 2. 100 g n 2 x 1 mole n 2 280 g n 2 0357 moles n 2 have 100 g h 2 x 1 mole h 2 202 g h 2 495 moles h 2 have The reactant that produces the least amount of product is the limiting reactant. How_to_determine_limiting_reactant_given_moles 13 How To Determine Limiting Reactant Given Moles Book How To Determine Limiting Reactant Given Moles Limiting Reactant and Percent-Lifeliqe 2019 This lesson plan covers analyzing chemical reactions in order to determine limiting reactants and excess reactants including calculating the amount of excess reactant.
Find the limiting reagent by looking at the number of moles of each reactant. The key is to keep the same reactant on top as the step above. Completing the problem using the moles have of the limiting reagent.
Balance the chemical equation.

How To Solve Stoichiometry Problems With A Conversion Box The How To How To Videos Watch And Learn Chemistry Worksheets Teaching Chemistry Chemistry Help
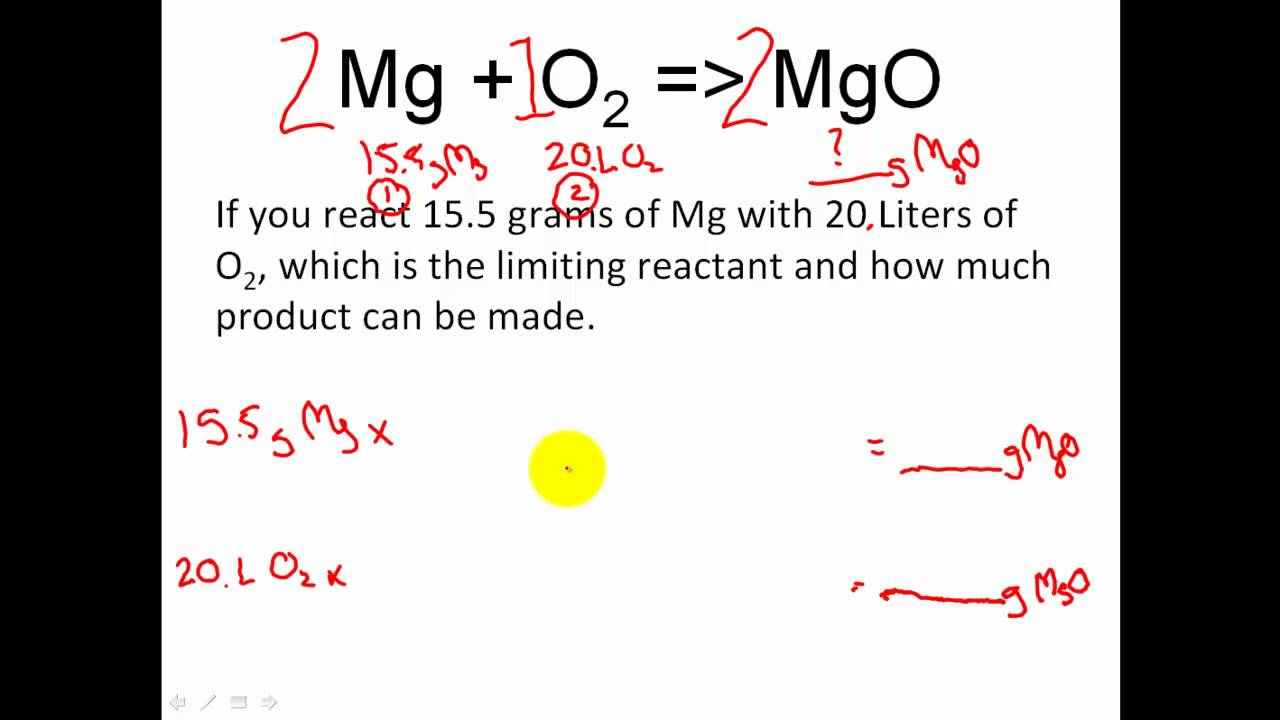
Stoichiometry Limiting Reactant Excess Reactant Stoichiometry Moles Module 6 Learning Psychology Apologia Chemistry School Work

This Particular Video Works Really Well With Apologia Chemistry Stoichiometry Tutorial Teaching Chemistry Stoichiometry Chemistry Apologia Chemistry

How To Calculate An Empirical Formula Chemistry Worksheets Teaching Chemistry Chemistry Help

Equilibrium 2 Calculating Equilibrium Teaching Chemistry How To Learn Chemistry Chemistry Lessons

Aluminum Foil Limiting Reactants Stoichiometry Chemistry Lab Percent Yield Mod 8 Stoichiometry Chemistry Chemistry Labs Teaching Chemistry

Stoichiometry Made Easy The Magic Number Method Chemistry Lessons Chemistry Help Chemistry Classroom

How To Calculate Percent Yield In Chemistry Teaching Chemistry Chemistry Physical Chemistry

Limiting Reagent Chemistry Tutorial Youtube Chemistry Tutorial School

Limiting Reagent Theoretical Yield And Percent Yield Youtube Chemistry Help Chemistry Teaching

Limiting Reactant Example Problem 1 Chemistry Problem Fuel Cells

Excess Reactant Unit 1 Stoichiometry Com Imagens Quimica

Stoichiometry Problems Solved Moles Problem Solving Boyle S Law Solving

Stoichiometry Moles To Grams Youtube Teaching Chemistry Chemistry Classroom Science Chemistry

What Is Stoichiometry And Why Is It Used In Chemistry A Plus Topper Whatisstoichiometry Stoichiometry Chemistry Chemistry Chemistry Worksheets

Balance Different Types Of Reactions With A Simple Yet Efficient Method To Help Avoid All The Confusion M Chemistry Worksheets Teaching Chemistry Mcat Study

Limiting Reactant Practice Problem Science Sciencewithtylerdewitt Tylerdewitt Tutor Sciencehelp School Help Online School Chemistry

Need To Calculate The Limiting Reactant Of A Chemical Reaction Chemical Reactions Chemical Chemical Equation

Post a Comment for "How To Determine Limiting Reactant Given Moles"